Mining and smelting Iron Ore and other metals
Metal ores are mined in a variety of ways, lead, tin and zinc have all been recovered using drift and shaft type mines whilst open cast mining is a feature of British iron ore mining.
Iron Ore Mines
By the time the railways came along iron ore was the most important mineral being mined. British iron ore deposits are mainly found in the North of the country, which coupled with the similar dispersion of coal reinforced the trend for heavier industries to develop in those regions. In the period between the two world wars the iron and steel industries migrated to Lincolnshire and Northamptonshire where large new steelworks were erected.
Fig ___ British Iron Deposits
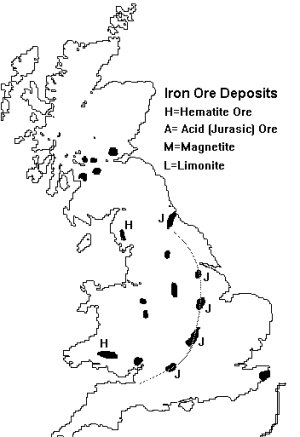
British ores tend to be of indifferent quality however, so there has always been a significant import of higher quality ores from abroad.
The most common ore type in England is a dark ore commonly called Spathic, the technical name is Siderite and chemically the material is iron carbonate. Siderite is often found mixed with either clay or coal deposits, mixed with clay it is called ironstone, when mixed with a coal deposit it is called blackband ironstone. Siderite is a poor quality ore, in Britain typical specimens contain just over thirty percent iron. This kind of ore forms deposits of so called 'Jurassic' iron ore found in a chain along the northern face of the limestone escarpment running from Bristol via Banbury, Kettering and Scunthorpe to Cleveland. These ores are acidic, that is they contain phosphorous, and could only be used after 1879 when Sidney Gilchrist Thomass came up with the idea of adding limestone to the smelting furnace. As about half the iron ore on earth is contaminated with phosphorous this virtually doubled the worlds potentially available steel.
The most valuable of the Jurassic ores is 'Brown Haematite' or 'Limonite', which is actually iron oxide,and British ores contain about forty percent iron. These ores also contain phosphorous and water (in varying quantities) and one major source is Northamptonshire. Limonite is yellow in colour, hence the name, and has been used as a pigment (yellow ochre) for thousands of years. Where this ore is found mixed with clay it is usually a dark colour and is called Ironstone (hence ironstone hoppers on the railway). The main source of ironstone is at Cleveland but it was also mined as far afield as Seend near Devizes in Cornwall. As the ore lies close to the surface a form of strip mining has long been employed.
First diggers remove the top soil and place this to one side. Next the soil lying over the seam of ore is removed, this is usually used to fill in an already worked area. The top soil is then put on top of this and the land returned to agricultural use. Meanwhile coal in the form of poor quality odds and ends with a large proportion of dust (technically called 'slack) is dumped on top of the exposed ore. Diggers then work their way along, picking this mix up and dumping it behind as they go. The mixed iron ore and coal is set alight and the coal calcines the ore. Once the ore is cool enough a third digger puts it directly into railway wagons.
This method results in a pleasing modelling arrangement as shown below which being long and thin (a minimum of only four or five inches) has potential as a screen in front of a hidden fiddle yard or hidden return loop.
Fig ___ Ironstone mining in Cleveland area
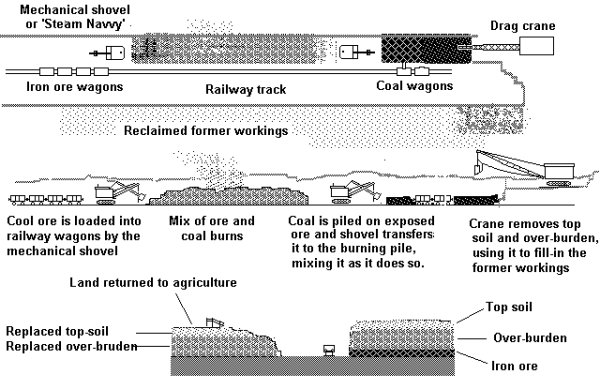
For exhibition purposes the mechanical shovels and drag crane could be made to move and you could add a smoke unit under the burning mix of ore and coal. The railway line serving the cutting would normally be worked by a works loco, in most cases the line would feed direct to the steel works. If the ore was to be shipped to a remote blast furnace exchange sidings would allow the railway company loco to collect and deliver the wagons.
Magnetite ore, which is dark reddish grey to black, has been mined in the West of England but the deposits were small and soon exhausted. Magnetite is the richest iron ore with a typical iron content of up to sixty percent. Magnetite is of course the 'lodestone' used in early magnetic compasses, it is named after Magnes, a Cretian shepherd who according to legend found the nails of his boots stuck to the rock on which he was standing. Britain has been importing quantities of magnetite since the 1930's at least (possibly before), mainly from Sweden and Norway.
Of the remaining ore deposits occurring in the UK the most valuable is Haematite otherwise known as Red Ochre and chemically iron oxide. This ore was the best in Britain and contains up to 20% iron, as a comparison the yellowish Leicestershire ore contains only 2%. Unfortunately Haematite was only found in the area of Barrow in Furness where the ore is dark grey to black, and Glamorgan where the ore is reddish brown to grey. Deposits in both these areas were exhausted by about 1960. Similar reddish brown ores in the Forest of Dean were worked out some time before the Second World War. By the early nineteenth century Britain was importing quantities of brownish red Haematite ore from the Bilbao area of Spain and these imports have continued.
Since the early 1970's imported iron ore has often been shipped in 'pelletised' form. The iron ore is ground to a powder and mixed with water, the resulting mix is passed to a balling mill which rolls it into small balls about the size of large marbles. These are dried and shipped in bulk, they are reddish brown in colour.
In order to make iron and steel alloys there were considerable imports of related ore types, notably Chromium, Manganese, Molybdenum, Nickel, Tungsten, Cobalt and Vandium. After the Second Word War however the countries producing these ores began locally refining them and selling the much more valuable metals in ingot form.
The iron is smelted at large iron and steel works, these are considered separately in Lineside Industries - Iron and Steel Works & Stockholders.
Mining and smelting Non Ferrous Metals
Britain was the worlds largest producer on non ferrous metals up to the late nineteenth century when the mines went into rapid decline. In the 1860's Cornwall was producing nearly half of all the worlds tin, by the 1930's it was producing only about one percent. Copper production in the 1850's was running at over three hundred thousand tons a year, within ten years this was down by a third and twenty years later it was down to less then forty thousand tons a year, precipitating the collapse of the Swansea smelting industry. Lead also went into decline in the later nineteenth century but here the fall in production was not so rapid and lead mines continued to operate into the 1970's.
Fig ___ Non Ferrous Metal Mining Areas
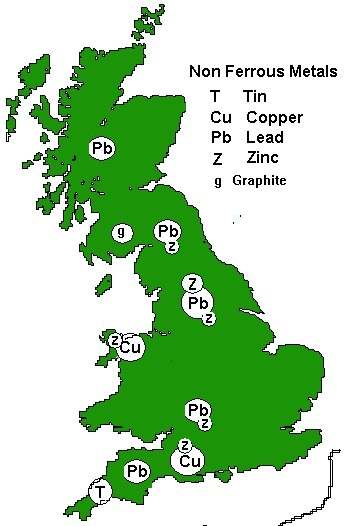
Lead
Lead mines once proliferated in the limestone areas of Britain, mainly Derbyshire, Somerset and the Yorkshire Dales, the main production area was the Peak District (Millclose was the nation's largest lead mine, it closed in 1939), however lead is found (and has been mined) in all the highland areas of the country. At the end of the nineteenth century over half the worlds lead was produced in Britain but by the 1960's lead mines were only in operation at Alston Moor on the Cumberland/Northumberland border and in North Derbyshire. Notable lead mining areas were located at Crooked Oak in Southern Northumberland, Newborough also in Northumberland, and in South Wales in the counties of Glamorgan & Gwent (Nant y Mywyn mine at Llandovey in Dyfed was the biggest lead mine in South Wales, opened in the late eighteenth century it finally closed in 1932). Millclose in the Peak District was the nation's largest lead mine and only closed in 1939. After the lead mines closed there was a fair bit of re-working the waste material from the spoil heaps to recover fluorspar (calcium flouride used in steel making, glass manufacture (and in place of glass for some specialist camera lenses) and the production of hydrofluric acid. Blue John is a type of fluorspar), barytes (barium sulphate, used in paper making and in the manufacture of paint as well as as an additive to cement) and calcite (calcium carbonate, used in cements, mortars, glass making and for ornamental purposes).
Lead is mined in the form of Galena, naturally occurring lead sulphide (PbS) which is appropriately enough lead grey in colour and usually associated with limestone deposits. The ore is sometimes called 'backstone' in older texts. Early lead mines were shallow affairs but by the 17th century deeper mines were the norm as the surface deposits were worked out. This meant that water became a problem in many mones and adding a drainage tunnel or 'sough' was common practice, later supplimented by horse 'gins' and steam pumping engines.
Once the ore has been mined the lead has to be extracted, or smelted. Step one is to dress the ore, basically break it down to about pea size, then crush it to a powder. The powder is then mixed with water to separate the heavy ore from the lighter waste materials. This was done in various ways for details of which see also Mines - General introduction, it is worth noting that in lead mines this work was often done partly underground, where the water being drained into the sough could be employed and as a result less waste was lifted up the shaft. If your model is set after about 1870 the crushing and separation was done in a large buildings, driven by steam or a water wheel. One large water wheel remains at Park Level Mill at Killhope in the Pennines, when it was set up in the 1870s there were many lead mines and many water wheels in the area. Killhope is the most complete lead mining site in Britain and restoration of the machinery is continuing.
Lead mines often did not have the substantial pit head gear associated with coal mines, it was not unusual for a single engine house to operate several shafts, with the lifting wires arranged on a series of grooved wheels mounted on short (9 foot) supports. Any pit type mine would be likely to require a pump (where an adit or drainage tunnel could not be laid in), one of the more common early steam types was the 'Cornish' pump engine, these large beam engines were used all over the country not just in Cornwall. They remained in use in the 1930s, although alternatives such as electric pumps were by that time available, see also Mines - General introduction.
To extract lead from galena (Lead Sulphide, PbS) the crushed ore is heated with a limited supply of air, this was done in a brick furnace, broadly similar in appearance to the common beehive type bound with iron bands and associated with a tall chimney. Once the mix has been sufficiently heated the air supply is then cut right off. The net effect of this is that the sulphur is driven off as a gas, mixed with oxygen from the early part of the process (most mines simply vented this to atmosphere). The unwanted muck floating on top of the molten metal would be scraped off then the lead would be drained from a tap hole.
Small mines used a small hearth to melt the ore, requiring nothing more than a substantial building with a chimney. In the late 18th century the London Lead Co developed a variation on the reverbatory furnace (which has fuel on one side, ore on the other and a domed roof to reflect heat from one to the other) which soon became the most common way of melting the ore at larger works but by the end of the 19th century the blast furnace was increasingly used for smelting the lead. Photos are hard to find, the sketch below is based on a tracing I made some years ago from a rather small photo in a book. The furnace (the circular tower on the left) appears to be clad in concrete, the tower on the right appears to be clad in either vertical wooden planking or corrugated iron sheeting (or so it says on my notes). The purpose of the chute on the right of the tower was not apparent, it may however have been used for loading mine spoil into railway wagons. The narrow gauge tracks at the bottom bring in tubs of prepared ore to the tippler (the elevated timber 'shed' at the base of the tower housing the mechanism for this)
Fig ___ Sketch of a lead smelting plant

In the blast furnace the ore is mixed with coke and limestone and the slag and molten lead is drawn off at the bottom as with steel. One difference however is that the air blast pipes are attached just above the base crucible, so they blow across the top of the slag and molten lead. The sketch below was prepared at the time I made the tracing, it is a rough drawing showing the dimensions estimated from the photo.
Fig ___ Rough sketch of smelter showing approx dimensions (British N)
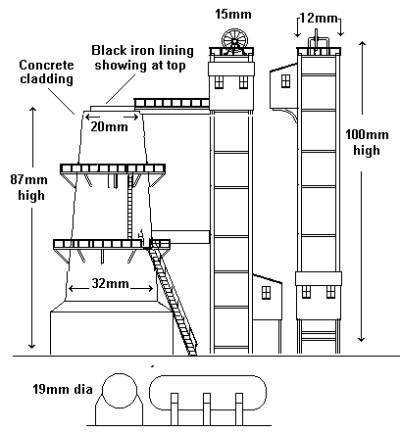
There are usually impurities in lead which require removing, some such as silver actually make this a profitable exercise (quite a few mines were named 'lead and silver mine' and some galena is mined primarily to recover the silver). A lead smelting works would not be located close to a town after the mid 18th century but would probably be rail served.
The lead was run off into sand moulds where it formed ingots or bars called pigs, typically weighing in at about 130 lbs (about 60Kg). Lead is heavy stuff, a pig about four inches square would be about eighteen inches long, a ten ton wagon carries 10200 Kgs so would hold 170 pigs of lead. This requires very little height and at least one lead smelter in Wales operated some low sided wagons, as the lead is valuable these had a pitched roof. After several house moves I was not able to find the photograph, the sketch below is from memory and may be inaccurate in detail (an amended drawing will be added when the photograph has been found). A model could be made easily using the Peco 'salt wagon', simply cut the sides down to three planks in height and fit the roof, the remaining 'four plank' frame can be used to make a four-plank wagon (on a second Peco chassis) or cut down and fitted with a roof from a second salt wagon kit on a spare chassis (you need the coupling plugs from the chassis kit), this leaves you with a standard seven plank wagon body and chassis.
Fig ___ Lead company PO wagon (provisional drawing)
At some works the lead was further processed on-site, usually to produce rolls of lead sheet, but a lot was shipped off to other works for forming into sheet, tubing etc. Lead being exported was shipped in rolls, about 3 feet long by a foot in diameter and it seems likely that this was a standard size for inland traffic as well.
Lead smelting works close to the coast sometimes imported the ore for processing (notably from France and Germany).Notable lead smelting works were located in Wharfdale, Nidderdale, Airedale, Craven, Rossendale in Yorkshire whilst Swaledale and Wensledale (again in Yorkshire) became important in the nineteenth century. Other works were located at Hengistbury Head Dorset, Combe Martin and Here Ferrers in Devon, County Durham and Teesdale. Lead smelting in Derbyshire started in about 1570 using a simple ore-hearth bellows fed then coal fired reverbaratory or cupola in about 1737. Smelters in the upper Dove valley around Greenlowfield, Mill Dale, Ellastone and Crakemarsh produced lead, zinc and copper.
Lead has many uses, it was added to glass to make 'lead crystal', it was used in pottery glazes, more recently it has mainly been used for making motor car batteries and in the manufacture of TEL (Tetra Ethyl Lead) the anti-knock agent that was used in petrol until the 1990s.
White lead is lead carbonate, it is used in paints (this is the paint that turns black on exposure to sulphurous pollutants in the air), up to the 1960's about a fifth of the lead used in Britain went into paint in this form. It is made by allowing a lead sheet to oxidise for several weeks in a warm atmosphere containing water and ascetic acid (vinegar), the white powder forms on the surface and is scraped off.
Red lead is lead oxide (Pb3O4) produced by heating lead in contact with air. Red lead is used in glass manufacture and mixed with linseed oil as a cement for joining lead pipes which were used for domestic water and gas up to the 1970's. Lead oxide comes in various forms however and the reddish brown form known as Litharge is used in making lead glass.
Lead has been used for lining sheaths on electrical cables as it does not rot, about a fifth of all the worlds production was used in this way prior to the development of petrochemicals and plastics in the 1960's. The third major use is of course in batteries such as those used in motor vehicles.
A tin-lead alloy was used for toothpaste tubes and similar, the mix being slightly different from that used for solder.
Lead was also widely used for sheathing electrical cable,
Other minerals associated with lead, such as fluorite, baryte and calcite, were traditionally regarded as waste but in the post war era these have proved valuable (lead is no longer mined in the Peak District but fluorite is an important commodity).
Copper
In the 1850's Britain produced about half of the worlds copper, most came from mines in Devon and the inland riverside port of Morewellham was, in Victorian times, the worlds most important copper port. This port was abandoned when the industry slumped toward the end of the nineteenth century but was partially restored in the 1970's as an open air museum. There were copper mines elsewhere however, for example there are some remaining buildings at Ecton in Staffordshire and there were a number of productive mines in Cumbria, although the import of cheap foreign ores had killed virtually all British copper mines off by the early 20th century.
By the 1960's 40 countries were between them producing over three and a half million tons of refined coper a year but 80% came from the USA, Chile, Zimbabwe (then still Rhodesia), Russia, Canada and the Congo (in that order).
Copper ores come in a range of colours; Chalcopyrite (Copper Pyrites CuFeS2) is yellow, Bornite (Cuprous iron sulphide) comes in many colours, it is known as peackock ore, Cuprite is Cuprous Oxide and is ruby red, Malachite (basic carbonate) is bright green, Azurite (also a Basic Carbonate) as its name suggests is blue and Copper Glance is the reddish brown Copper Sulphide.
British copper ore is usually of the Glance type. In the mid 1850s William Henderson, of Glasgow invented a simple methood for smelting copper by roasting the burnt pyrites with salt and washing out the copper with water. In more modern smelters the ore is crushed and ground in water, pine oil is then added and the mixture churned up, a foam forms on the surface containing most of the copper iron sulphide which is then skimmed off. Smelting follows a preliminary roasting, limestone or silica (sand) or both are then added and the mix is heated in a reverbatory furnace, the iron forms ferrous silicate slag floating on top which can be skimmed off. The remaining mix, known as 'matte', is placed in a vessel similar to a Bessemer converter and air is blown through it, this results in pure copper being left behind whilst sulphur dioxide is given off as a gas. There is some sulphur dioxide left however, causing blistering on the surface of the copper ingots, which for this reason are called 'blister copper'. Blister copper is 98% pure but electrolytic reduction can improve this to 99.99%.
With the growth of imported copper ore there was a trend to locate new works close to coastal towns, although there was also a trent toward importing copper in ingot form, refined in the country of origin.
Copper is today the second most widely used metal after iron, it is second only to silver in its ability to conduct electricity and heat so it is used in boilers, condensers, radiators and the like as well as in electrical wiring and other equipment. Today over half the copper produced goes into electrical equipment, copper wire containing manganese has a high electrical resistance, it is called 'resistance wire' and is used to make the 'elements' for electric fires and toasters. Copper is used as an exterior cladding on buildings in the form of thin sheets nails down, it is more resistant to corrosion than iron as it forms a green coating (of hydrated basic carbonate) on the surface. The dome of St. Paul's cathedral in London, and the Statue of Liberty in New York are both coated in copper sheet and both a light green colour. Copper can be beaten thin enough to allow light to pass through (about a five hundredths of an inch). Copper is used in alloys such as brass (copper and zinc), bronze (copper and tin), 'copper' coins (copper and tin) and 'silver' coins (copper and nickel).
Zinc
Zink is often found in the same areas as lead, notable Zinc mines include a large establishment at Batws Y Coed operated by the chemical company Brunner Mond up to 1906. The mine re-opened for a time after the First World War but was not a success. Zinc was used as a metal and also for coatings on other metals. Brunner Mond wanted zinc sulphide for use in making chlorine from the calcium chloride produced at their soda ash plant at Northwich (Cheshire). Zinc is often found close to lead and was exploited by lead mine owners, always in need of additional income. There are large deposits of Zinc on the island of Anglesey, however these have never been fully exploited.
Although brass had been known about for centuries it was only in the 17th century that the Chinese found out how to obtain pure zinc, and it was in the 1780's that someone in Bristol worked out the method (the key was keeping the process out of contact with air). The ore is separated in a foam bath of water and pine oil as for copper, the skimmed froth is then roasted, mixed with coke and heated to a high temperature in clay retorts. The zinc boils
off and is concensed into iron buckets. Zinc is used for making brass (7 parts copper to 3 parts zinc), bronze (eight parts copper, ten parts tin with two parts zinc) and for 'dry batteries', the majority of the metal however is used in coating iron to produce 'galvenised iron'. The iron sheets are cleaned then dipped in molten zinc. Zinc Sulphate (ZnSO4) is used with water to produce 'white vitriol' and when this is mixed with barium sulphate you get a white powder precipitate. This is used in white paint, it does not cover as well as white lead but it does not turn black on
exposure to polluted air (this was used to good effect by the Midland Railway, the markings on their wagons being noted for not darkening as muchj as those on other companies goods stock).
Tin
Tin is usually mined in the form of 'tinstone' or Cassiterite, the ore is almost always stained with iron and may be yellow, reddish brown or even black.
The name Cassiterite comes from the Greek word for tin, in the Bronze Age Britain was known as the 'Cassiterides' or 'tin islands'. Up to about 1860 Cornwall was the worlds largest producer of tin (in Victorian times about a third of all the tin used in Europe was produced in Devon and the Taymar valley), but by the 1960's only two mines remained; South Crofty at Cambourne and Geevor at St Just (near St.Ives), the ore from both was shipped to Bootle in Lancashire for smelting. These two mines produced only about a thousand tons of tin a year and we imported a further twenty five thousand tons a year to support the tin-plate industry in South Wales. The last Cornish tin mine at Geevor ceased operating in 1990 and is now a heritage museum.
The Poldark Mine & Heritage Complex, Wendron, Helston TR13 0ER (Tel: 01326 573173) includes an eighteenth century tin mine along with an associated museum XXX contact for more info XXX. There is a superb model railway layout called Wheal Louise, built by XXX it depicts a Cornish tin mine in the nineteenth century. This layout has featured in Railway Modeller Magazine (XXX dates) and is worth studying for anyone contemplating something on similar lines. Tin was also mines on dartmoor.
The ore is crushed, washed and roasted to remove impurities such as arsenic and sulphur. The remaining ore is then mixed with anthracite coal (almost pure carbon) and heated, which gives a fairly pure metal. This is then poured
onto a sloped surface, the metal runs down leaving impurities behind. At this stage the liquid metal is stirred with green wood poles XXX see existing notes. The major use for tin is of course tin plating iron or steel.
Chemists call tin by its Latin name Stannum which gives its chemical symbol Sn. Tin ore (cassiterite) is crushed, washed and roasted to remove impurities such as arsenic and sulphur. The remaining ore is then mixed with anthracite coal (almost pure carbon) and heated, which gives a fairly pure metal. This is then poured onto a sloped surface, the metal runs down leaving impurities behind. At this stage the liquid metal is stirred with green wood poles the moisture in which causes violent boiling bringing the impurities to the surface where they can be skimmed off.
Tin can be rolled into very thin sheets and although metal foil is nowadays mainly aluminium it is still commonly called tin foil. Tin is too expensive to be used in pure form for food wrapping and up to the 1960's tin and lead either as an alloy or in the form of lead sheet with tin foil pressed to each side was used.
Tin is a useful coating material forms the basis of several alloys. Tin melts at 232 deg c, lower than most other metals. Tin and lead make solder which actually melts at a lower temperature than either of the two ingredients, Woods Metal is a tin lead alloy that melts at only 66 deg C, it is often used for sprinkler systems. Brass and bronze are both alloys of copper involving tin, these are discussed below.
We often use the word 'tin' when speaking of 'tin plate', which is iron or steel sheet plated with tin, the best example being the tin can. To distinguish things that are actually made from tin the term 'block tin' is usually used for pure tin, tinplate for the coated materials.
Tin-plate
The British tin-plate industry was mainly concentrated in South Wales, where iron works existed and Cornish tin was easily available. The tin plate industry in Swansea dates back to at least 1665 when Cornish tin was used to coat iron sheets but it was the eighteenth century before industrial scale production was developed (the first factory opened in Ponty Pool in the 1730's). In the later nineteenth century demand increased with the increasing use of cans for food and one gallon tins for petrol and this was in part facilitated by the introduction of steam powered rolling and stamping mills. The Midlands also had a substantial tin plate industry and Britain built up a major export trade in tin-plate, a lot of tin plate was used for roofing in America. Originally the industry used Cornish tin but as this began to run out in the 1860's tine was imported from Australia and Malaya. In the 1880's and 1890's steel replaced iron for tin plate (usually Seimens rather than Bessemer steel).
Tin plate was made by dipping iron or steel sheet into liquid tin. To prepare the metal for plating it is first dipped in palm oil recovered from Palm kernels by seed crushing firms and shipped in drums or small tank wagons. The oil ensures a smooth and even coating on the sheet. The seed crushing industry is discussed elsewhere. Modern tin-plate, as used for food cans is actually steel, supplied to the tin plate works as coils of thin sheet it is given a thin coating of tin by electroplating.
Although we use tin plate for coating food storage containers it is worth noting that most of the alloys of tin are toxic, especially the organic ones.
A tin-plate works would receive consignments of sheet metal, prior to the 1950s this material was delivered in small batches, cut down to the size a man could conveniently handle (about three foot square). The metal might be delivered as coils of strip metal from the mid 1950s on, although only at a larger works, delivered on converted (roll) wagons.
Tinplate works would ship out mainly van loads of finished tinned sheet, typically in small stacks of sheets about three foot square. Several firms operated their own railway vans in the pre-grouping era, I believe some remained in service into the early 1930s.
Fig ___ Tin-plate manufactures vans
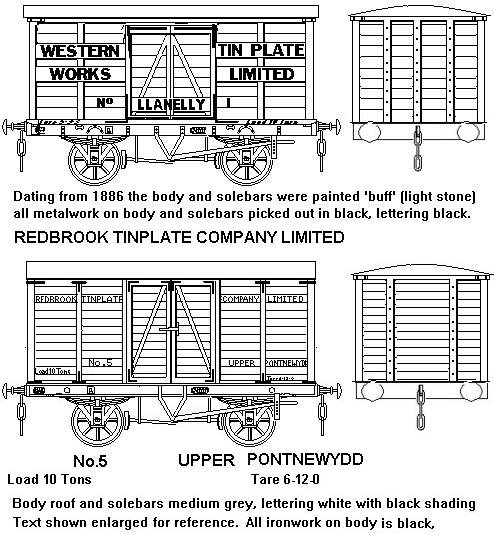
The lower example is sketched from a photograph in Peter Mathews' book 'Private Owner Wagons' which is` detailed in the bibliography.
In photographs from the mid 1950s I have seen pale wooden boxes about three foot by two foot by about ten inches deep with three inch square batten on the base to allow fork-lift handling.
Tin boxes and cans
In Britain tin boxes were used for a range of products, the makers relied on tins for cigarettes and the square boxes used to sell bulk biscuits in small shops for their bread an butter. Tin boxes were mainly hand made and often quite complex in design as their prime function was to advertise their contents. Early tin boxes were decorated either with paper labels or with more expensive 'transfers' but in 1875 'offset lithography' printing was invented and a tin maker by the name of Fry bought the patent.
Offset litho printing is quite straight forward, a flat master plate is prepared with a raised pattern of the image to be printed, this is then covered in ink. The original process used a sheet of cardboard to pick up the ink from the raised portions of the plate and transfer this to the tin or paper being printed. Later the cardboard was replaced by a rubber roller.
The main use for this printing system was the complex printing of cheques for banks, one of the main customers was Barclays Bank and a Mr. Barclay went into partnership with Mr. Fry.
Barclay & Fry used their patented process to print paper as well as tins and in 1922 they became the core of one of the early 'big business' concerns in Britain, calling themselves 'Metal Box & Printing Industries'. The printing side remained important, accounting for about half the profits of the firm in the 1960's.
The tobacco companies were big users of tins but sealed tins could not be used as opening the tin damaged the cigarettes. In 1888 a Mr. Williamson, head of the company, patented a simple cutter which could be built into a tin lid. This allowed the tin to be sealed with very light metal, twisting the lid operated the cutter, removing the top neatly. This proved a boon to the cigarette firms, notably the largest firm in Britain Rickets Wills & Co (which became WD & HO Wills).
Preserving food by bottling and heating using air tight glass containers was invented by Franscois Appert, a Parisian confectioner, in 1810. He developed the idea in response to a reward offered by the French government who were looking at ways to feed the army in the field. A few years later an Englishman by the name of Peter Durand developed a system based on metal containers and tinned foods appeared in the shops by 1820. Tins recovered from an 1824 expedition to the Arctic were opened in 1911 and the food inside was found to be still in good condition after 87 years. Tin cans were all made the same way, a three piece assembly consisting of a strip to form the tube and two disks for the ends, these were soldered together to make the tin.
The 'key' opening tin can was developed in America in 1867 but the can opener for ordinary cans was not commercially available until about 1914 (people used a sharpknife to puncture the lid and work their way round the edge cutting through the metal).
In Britain tin cans remained a small scale business, people bought them empty and filled them at home with food to be preserved. In America the extingencies of the American Civil War forced development of industrial canneries and the American Can Company soon became the worlds largest supplier. The tins were delivered already formed into a tube and with the bottom plate in place, the contents were added, the lid soldered on and the whole thing cooked or 'pasteurised' to kill any remaining bacteria.
Some firms tried building assembly plants close by the consumer, this saved on shipping costs as the tins were shipped flat, packs of sides and packs of lids and bottoms. The bulky completed tins with separate lids were then shipped the short distance to the customer.
Meanwhile American farmers began producing foods specifically for canning, in Britain tins of Californian fruit were a common commodity by the 1920's but British canning was still mainly concerned with home preserving of seasonal foods on the basis of 'Eat what you can and can what you can't eat'. In the later 1920's the Americans began looking toward expanding in this country and established the British Tin Company.
Metal Box and Printing Industries saw this as a threat and had to work out a deal with the American firm Continental Can Company to set up in competition, in the end they forced the Americans out of the British can market.
At the cannery, usually located close to where the food is produced as it must be fresh, the food is washed and inspected and sorted for size and quality. It is then peeled, shelled, boned, blended or whatever and then cooked. As some foods shrink with cooking they are cooked before they are put into the cans, other foods are heated in the tin.
The tins should be well filled but the filling machine sucks any remaining air out of the tin and seals the lid. The sealed tins are then placed in boiling water or heated with steam to kill any bacteria inside the can and when cool the labels are added. The cans of corned beef from Argentina which caused outbreaks of food poisoning in the early 1960's were found to have small pin holes in the seals, they were cooled by placing them in nets and dipping them in the river Plate, from where bacteria entered.
Tin can manufacturing, and the food canneries they supplied, both benefited from a rail connection for delivery and despatch.
Cans did not have it all their own way however, the re-usable glass bottle, although inconvenient for the consumer, was cheaper for the supplier and it was easier to open than the traditional three piece can (consisting of tubular body, top and bottom). In 1963 the pop-tab opener appeared, the first successful design coming from the American giant Continental Can. This provoked a massive increase in the use of tins for beverages such as beer and soft drinks, ushering in what the industry called the 'era of the six pack'. This in turn pushed the development of the non-returnable one-way glass bottle, which uses much thinner glass and so is cheaper to produce.
Research continued and in the 1970's the Aluminium two-piece can, made from a one piece formed body and base with a separate lid came into play. This was proved popular with the beverage industry and because of the cost of the aluminium recycling is worthwhile, a recycled can uses up 89% less energy in manufacture than a new one.
Bronze
Is an alloy of copper and tin, it wears well and is used for machine parts and name plates on buildings. The usual mix for common bronze is eight parts copper, ten parts tin with two parts zinc but there are a number of types of bronze each with different constituents and used for different jobs.
Gun metal is a form of bronze used for steam and water fittings.
Manganese bronze, made from copper, zinc, tin and manganese is used for casting ships propellers because it is not eaten by sea water (see also Lineside Inductiries - Scrap metal Yards, Foundries and Forges).
Brass
Basic brass is an alloy of copper and zinc, usually including a small amount of tin, it is not as strong as bronze. The usual mix is seven parts copper to three parts zinc but there are many kinds of brass, yellow brass has lead added and 'Naval' brass has more zinc than tin. Brass does not rust, so its used in pumps and plumbing fittings, but it does tarnish so it is often plated for domestic uses. Electric furnaces are usually used to make the stuff and it is then cast at a foundry to produce salable goods (see also Lineside Inductiries - Scrap metal Yards, Foundries and Forges).
Aluminium
Aluminium is the most common metal, accounting for about 8% of the earths crust, unfortunately it is found in highly stable compounds and is difficult to separate out as a pure metal. In the time of Napoleon III aluminium plates were reserved for the emperors table, the lesser nobles had to make do with gold and silver. The first British Aluminium Foundry was set up by Sir William Mills at Sunderland in 1885 at which time Aluminium was a precious metal, with a value slightly higher than gold, the pricipal method of obtaining the metal was a complex chemical process.
The first patents for extracting the metal on a commercial scale using electricity date from 1886, at this time it was still very much a semi-precious metal, valued at over three pounds and ounce. The extraction process has two stages, firstly the Bayer Process is used to extract aluminium oxide (alumina) from bauxite ore. Next the Hall-Heroult process is used to extract the oxygen, leaving behind the pure metal. This process was independently discovered by Hall (1863-1914) in America and by Heroult in France but it was the American who set up the first commercial production company in 1888. The British Aluminium Company set up their Scottish works using the new processes in 1894.
Bauxite is named after the French town of Les Baux, where it was first discovered. Bauxite, which can contain between thirty and seventy percent alumina, comes in various colours depending on the impurities, it can be white, cream, yellow, brown grey or red. It is composed of aluminium oxide, iron oxide and water with traces of silica and titanium oxide.
To make one ton of aluminium you need 4 tons of bauxite, which is dissolved in a hot solution containing about 3 cwt of caustic soda (sodium hydroxide or 'lye'). The result of this process is a liquid which is filtered to remove unwanted oxides (mainly iron) and placed into tall cylinders, here it is 'seeded' with crystals of aluminium hydroxide and stirred. The aluminium forms more hydroxide on the crystals which grow and sink to the bottom of the tower. The crystals are then washed filtered and 'calcined' (cooked to reduce it to a powder) to produce the alumina (aluminium oxide). You get about 2 tons of alumina from the four tons of bauxite you started with, the caustic soda is recycled.
The Hall-Heroult process takes the two tons of oxide, adds a small amount of Cryolite to act as a catalyst and three quarters of a ton of carbon to serve as an anode. Cryolite is a compound of aluminium and sodium with a little fluorine, naturally occurring only in Greenland it is usually made synthetically.
You put this lot into a container lined with heat resistant brick and pass electricity through it, the electricity required takes about four to six tons of coal to produce. The molten aluminium collects at the cathode (mounted at the bottom of the chamber) and is siphoned off once every day or two.
The electrolytic process required considerable quantities of electricity and the British Aluminium Company (BAC, established in 1894) decided that hydroelectric power was the answer and so located their factory at Foyers beside Loch Ness in about 1900. A few years later they built another bigger plant (power station and smelter) at Kinlochleven and in the 1920s, a third and still larger operation was set up at Fort William. This latter factory is still operating in the early 21st century (now owned by Alcan) but the Kinlochleven smelter closed in the 1990s (the power station is still in use feeding the National Grid).
A new method, called the Toth process, has been developed which can recover aluminium from high-alumina clays such as kaolin (otherwise known as 'china clay'). This process uses much lower temperatures than the Hall process (only 260 degrees centigrade) and so requires much less electricity.
British Alcan has a large factory at Burntisland near Edinburgh to produce alumina from bauxite, but it also imports alumina powder via its private terminal at Blyth. Alumina is shipped in quantity in hopper wagons, the LNER and to a lesser extent LMS both built wagons for this traffic. British Alcan operated their own fleet of unfitted wagons in this traffic until the late 1980's and redundant grain wagons of the Peco fifteen foot wheelbase Grano type were also used in the 1980's. Alumina is used in making paper, abrasives, ceramics, light bulbs and motor engine spark-plugs as well as for producing aluminium.
Aluminium is used for structural engineering where its low weight is an advantage, it is also used in powder form in making protective paints for wood and metal. When the National Grid electricity distribution system was established light weight aluminium wire, wound on a steel core, was used for the power cables.
^
Go to top of page






